Stem cell therapy has shown itself as a supportive treatment for many chronic diseases, including Parkinson’s disease, dementia, ASD, multiple sclerosis, and stroke, among others. As patients explore this option, practical considerations come into focus—especially cost. For many, the question becomes: how much does stem cell therapy cost?
The answer depends on several factors, including the treated condition, treatment complexity, cell type, and regulatory standards of the country where therapy is provided. Costs can range from a few thousand to tens of thousands of dollars, and understanding what is included in that price is essential for making an informed decision.
What Does “Stem Cell Therapy Cost” Actually Include?
When evaluating stem cell therapy costs, it’s essential to break down what is included and what is not. Below are the main elements typically factored into the total cost.
Medical Evaluation and Diagnostics
Stem cell therapy begins with a thorough medical evaluation. This includes:
- Initial medical consultation
- Review of medical history and previous records
- Diagnostic evaluations, including blood tests
- Imaging studies, such as MRI or CT scans
These diagnostics help the medical team determine the appropriate treatment plan—deciding which type of stem cells to use, how many cells or injections might be needed, and whether multiple sessions are required.
Some clinics include the cost of these evaluations in their package price, while others charge them separately. Always clarify if initial consultations and diagnostic tests are included in the quoted price, as these can add significantly to the total if billed additionally.
Cell Sourcing, Processing, and Quality Control
A substantial part of stem cell therapy cost comes from harvesting the stem cells themselves. Depending on the origin of the cells, this may include laboratory processing, quality control tests, and storage.
- Allogeneic (donor) stem cells:
- Donor selection and medical screening
- Laboratory expansion and preparation of donor cells
- Safety, sterility, and viability testing conducted before and during administration
- Autologous (own) stem cells:
- Cell collection procedure, like liposuction or bone marrow aspiration
- Laboratory isolation and concentration of harvested cells
- Quality control and viability testing before treatment
High-quality clinics maintain GMP-grade laboratories and perform extensive quality control—ensuring cells are free of contamination and have the proper potency—which adds to the expense.
Find out more about the varieties, origins, quality, and safety of stem cells, as well as how they can be used to treat various long-term illnesses.
Read moreClinical Administration, Monitoring, and Follow-up
The treatment program brings together the core medical procedures and the care that accompanies them. Depending on the clinic and treatment format, it may include:
- Stem cell administration, delivered as a single or multiple injections/infusions over several days
- On-site medical supervision during and immediately after cell delivery
- Outpatient treatment or inpatient care within a multi-day program
- Clinic or hospital stay, including accommodation, nursing support, and routine medications
- Rehabilitation, like physical therapy or supportive treatments
- Follow-up consultations to evaluate progress after treatment, with a clearly defined duration or number of visits
Pro Tip: Ask the provider for a written list of what the stem cell treatment price covers. Does it include consultations, pre-treatment tests, the stem cell harvest or product, the procedure itself, post-treatment meds, and follow-up visits? If anything is not included, request an estimate for those as well.
Key Factors That Influence Stem Cell Therapy Cost
Several key factors drive the cost differences in stem cell therapy.
Medical Condition and Treatment Complexity
Generally, more severe or advanced conditions require more intensive treatment, which raises the stem cell injection cost.
For example, treating a simple orthopedic injury (like a mild tendon tear) might involve a single injection of stem cells and minimal additional therapy, whereas a progressive condition like late-stage multiple sclerosis might necessitate multiple rounds of stem cell infusions, higher cell doses, and extensive supportive care.
Type and Quantity of Stem Cells Used
The stem cell therapy cost is closely linked to the type of cells used and the dose required:
- Cell source: Treatments may use donor-derived mesenchymal stem cells (MSCs) ethically sourced after a healthy birth from placenta and umbilical cord tissue, with informed consent and medical screening, or autologous cells collected from bone marrow or adipose tissue, each involving different processing steps and costs.
- Cell quantity: Pricing may depend on the number of cells, vials, or injections included. Higher doses or repeated administrations typically raise the total cost.
Pro Tip: When comparing prices between clinics, check if they mention how many cells or injections are included in the stated price and whether additional administrations are billed separately, increasing the overall stem cell shot cost.
Number of Treatment Sessions
Stem cell therapy can sometimes be a one-time procedure, but often it involves multiple sessions to achieve the desired outcome. The required number of treatments will influence total cost. Each session will incur costs for medical care and possibly new cell preparations.
Level of Medical Supervision and Follow-up Care
Stem cell therapy is not just the moment of injection; it’s a program of care. The intensity and quality of the medical supervision you receive can influence the final stem cell treatment price. Some clinics advertise stem cell injection costs that only cover the injection and do not include diagnostics, monitoring, or aftercare. In contrast, clinics that bundle pre-treatment evaluation, on-site medical expertise, post-treatment rehabilitation, and follow-up support often provide clearer value and cost transparency than pay-per-service models.
How Much Does Stem Cell Treatment Cost Abroad: What Patients Commonly See
Average costs of stem cell treatment range from $5,000 on the low end to well over $50,000 on the high end.
| Most common price band | Reported prices most frequently fall between $10,000 and $30,000 for a single course of therapy. |
| Patient-reported spending (late 2025) | A patient survey showed the largest group paid $5,000–$10,000, followed by a smaller but significant group paying over $20,000. |
| Simple localized treatments | Procedures like a single joint injection with autologous cells may cost a few thousand dollars. |
| Complex systemic treatments | Multi-day or multi-infusion programs, especially for neurological or systemic conditions, often range from $20,000 to $30,000 or more. |
Please, keep in mind, that these are estimates, not quotes—actual prices will vary by clinic and individual case.
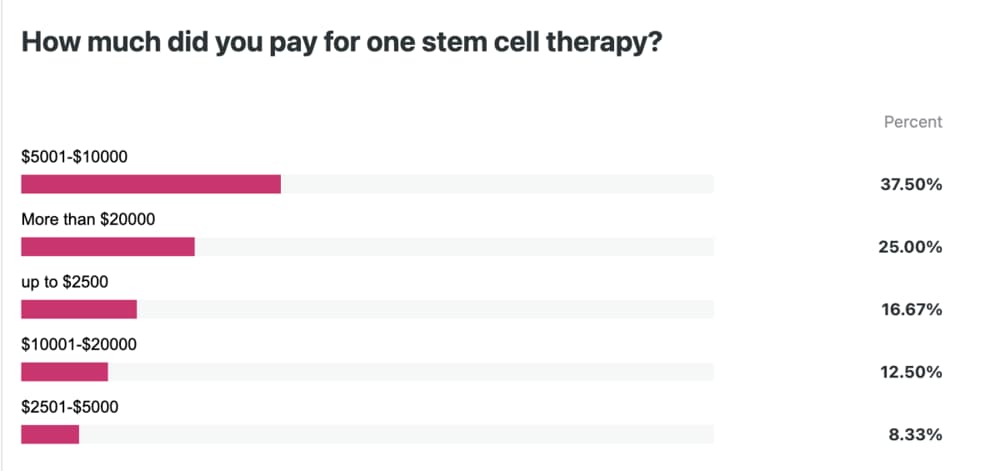
Stem Cell Therapy Cost in the United States
Stem cell therapy in the USA is shaped by strict FDA oversight—only a small number of cell-based treatments are broadly approved, so many MSC-style therapies are offered as experimental procedures with prices that often sit at the higher end of the global range.
Regulatory Environment and Pricing Structure in the U.S.
The FDA usually considers stem cell products as regulated biologics and states that the only approved stem cell products are blood-forming stem cells from cord blood for certain blood and immune disorders, not for general “regenerative” purposes. Because many treatments using MSCs are not standard or widely approved, pricing usually is set by the clinics themselves and may depend on the type of procedure or the number of injection sites.
What You’ll Often See in U.S. Quotes
A common breakdown looks like this:
- Initial consult + imaging: $800–$2,500
- Cell harvest: $5,000–$12,000
- Stem cell administration: $3,500–$9,000 per site
- Long-term follow-up package: $600–$1,500
What’s Typically Included in U.S. Stem Cell Treatment Costs—And What Is Not
Many clinics bundle the core procedure and a short follow-up window, but imaging, extended lab work, and rehab can be billed outside the headline price—so the only fair comparison is an itemized list of what’s included.
Stem Cell Therapy Cost in the United Kingdom
Stem cell treatments in the UK, as in the United States, are divided into two categories:
- NHS-funded therapies that are already established
- private self-pay regenerative services that are typically accessed outside routine NHS care.
Private Clinics vs. Experimental Settings
Private clinics operate under strict oversight and mainly offer orthopedic or localized treatments. Costs of regenerative stem cell therapy are relatively high, often similar to the U.S.:
- Initial consultation: ~£200–£300
- Single joint treatment (e.g., knee): £2,000–£4,000
- Complex or multi-session therapies: £20,000–£40,000+
Clinical trials are another option, typically run by NHS hospitals or universities. These may reduce or eliminate treatment costs, but access is limited by strict eligibility criteria and study design.
What’s Included in UK Stem Cell Treatment Costs
Quoted prices typically cover the procedure itself and short-term follow-up. Often excluded and billed separately are:
- Imaging (MRI, X-ray)
- Blood tests or additional diagnostics
- Physiotherapy or rehabilitation
- Medications after treatment
Some clinics also offer cell storage (e.g., cryopreservation of MSCs or cord blood), which usually involves separate fees, such as an upfront cost plus annual storage charges. Travel, accommodation, and meals are not included for patients coming from abroad.
How Much Does Stem Cell Treatment Cost in Europe
Stem cell therapy in Europe shows wide variation in cost, driven by differences in national regulations, healthcare systems, and clinic operating expenses. Several countries allow certain regenerative treatments under clinical or private frameworks that differ from U.S. rules, which has made parts of Europe a common destination for international patients.
Why European Clinics Attract International Patients
| Factor | Key points |
| Clinical standards | Many European clinics follow established medical protocols and operate under higher regulatory and quality standards compared to less regulated markets. |
| Cost structure | Central and Eastern European countries offer comparable medical infrastructure at a lower cost due to reduced labor, tax, and operating expenses. |
| Treatment model | Some clinics use bundled programs that include diagnostics, treatment, accommodation, and on-site support in one price. |
| Geographic accessibility | Short travel distances and established medical travel routes make cross-border treatment relatively straightforward for international patients. |
Cost Variation Between Western and Central/Eastern Europe
Mesenchymal stem cell therapy costs in Western Europe are typically high and often comparable to U.S. pricing, especially for complex or multi-session treatments. These costs reflect facility expenses and regulatory compliance requirements.
Central and Eastern Europe generally offers lower pricing for comparable treatment scopes. Clinics in Serbia, for example, follow European-aligned medical regulations while operating at a lower cost.
| Country | Typical cost of stem cell treatment in Europe | Notes |
| Switzerland | €20,000–€50,000+ | High-cost healthcare system with strict regulation and premium clinic infrastructure. |
| Germany | €20,000–€40,000 | Many regenerative treatments available mainly through clinical trials or research programs. |
| France/Spain | €17,200 – €43,000 | Usually available through clinical trials or in selected hospital cases where it’s medically justified. |
| Serbia | €10,000–€35,000 | European-aligned medical regulation with lower operational costs, allowing more competitive pricing. |
Get a free online consultation
If you are considering stem cell therapy, leave your contact details, and we will arrange a free online consultation with Swiss Medica doctors, where you can get a clear explanation of the expected costs and the potential outcomes for your individual case.

Medical Advisor, Swiss Medica doctor
Stem Cell Treatment Cost in Other Countries
Outside of the United States and Europe, many patients consider stem cell therapy in Asia, Latin America, and other emerging markets. Prices may be lower than in Western countries, though some destinations prioritize advanced or premium care over cost savings.
| Region | Countries | Average cost (USD) | Notes |
| Asia | India, Thailand, South Korea | ~$2,000–$30,000 | Wide range: lower-cost providers alongside advanced trial-based centers; regulatory oversight varies. |
| Latin America | Mexico, Panama, Colombia | ~$3,000–$20,000 | Popular with North American patients; generally lower costs and more permissive frameworks than the U.S. |
| Other destinations | Georgia, UAE, Maldives | ~$3,000–$50,000+ | From cost-focused medical tourism to premium and luxury offerings. |
Is the Cheapest Country the Best Choice for Stem Cell Therapy?
Lower prices can make stem cell therapy abroad appealing, but cost alone is a weak filter. The practical question is not where treatment is cheapest, but where safety, experience, and regulatory controls exist at a price you can justify.
Medical Quality vs. Price-Driven Decisions
Clinical standards matter more than the headline number. Factors like physician training, cell processing methods, infection control, and follow-up protocols directly affect outcomes and risk.
Some lower-cost countries offer strong care due to economic differences, but others rely on loose regulation. Evaluating published testimonials, clear information on the website, and treatment scope can help distinguish value from risk.
Travel, accommodation, companion costs, local diagnostics before or after treatment, and potential repeat sessions can narrow or erase apparent savings.
Learn what happens before and during treatment at Swiss Medica, who may be suitable for it, and how to improve the effectiveness of stem cell therapy.
Read moreStem Cell Therapy Cost vs. Expected Outcomes: Setting Realistic Expectations
In regenerative medicine, outcomes depend far more on medical factors than on the size of the invoice.
Why Stem Cell Treatment Cost Does Not Equal Effectiveness
What patients pay for is the treatment process—cell sourcing, laboratory handling, medical oversight, and supportive care—not a guaranteed biological response. Two clinics may use similar cell doses and protocols but charge very different prices due to differences in facilities, services, or overhead. Effectiveness depends on the condition being treated, disease stage, and protocol design.
Why Results Cannot Be Guaranteed
Stem cell therapy produces variable responses because each case is unique. Some patients improve significantly, others moderately, and some not at all. Ethical clinics avoid guarantees and frame therapy as a potential for symptom improvement or functional gains, not a cure. Patients should define in advance what level of improvement would justify the cost for them and proceed with informed, measured expectations.
Does Insurance Cover Mesenchymal Stem Cell Therapy Cost?
The question, “Is stem cell therapy covered by insurance?” is a common and critical one, given the high costs of treatment. In most cases, insurance (whether private health insurance or government plans like Medicare) does not cover elective stem cell therapies. There are a few exceptions, but they are limited.
Why Most Insurance Plans Exclude Coverage
Insurers generally classify regenerative stem cell therapies as investigational because they are not established as standard treatment for most conditions. Coverage is generally limited to treatments offered within approved clinical trials.
Limited Exceptions and Research-Based Reimbursement
There are a few exceptions where insurance might cover stem cell-related treatments:
- Established stem cell transplants: Insurance typically covers bone marrow or hematopoietic stem cell transplants for cancers and blood disorders.
- PRP therapy: Some insurers cover platelet-rich plasma for specific orthopedic conditions, though coverage is narrow and condition-dependent.
- Clinical trials: In registered trials, treatment costs are usually paid by the study sponsor, while insurance may cover routine care.
Some patients also use medical financing or health savings accounts for these costs. In the U.S., you can often use funds from an HSA (Health Savings Account) or FSA (Flexible Spending Account) for medical expenses not covered by insurance, including stem cell therapy.
Transparency and Ethical Pricing in Regenerative Medicine
When evaluating a stem cell therapy clinic, pricing transparency and ethical communication are often the clearest indicators of whether the provider is focused on patient care rather than sales. Here is what we recommend you pay attention to:
- Clear, written pricing: The clinic provides a detailed breakdown of what is included and excluded before treatment begins.
- Transparent cost structure: Fees are explained as a full program or with a realistic estimate of how many injections or sessions are needed.
- No misleading per-injection pricing: The clinic avoids advertising a low stem cell injection cost without explaining how many injections are typically required and what the total cost is likely to be.
- Written treatment plan and quote: Patients receive documentation outlining the proposed protocol and total cost prior to committing.
- No pressure tactics: Decisions are made after medical consultation, without time-limited discounts or sales urgency.
- Honest outcome communication: The clinic avoids guarantees and explains that results vary between patients.
What’s Included in a Stem Cell Therapy Program at Swiss Medica
Swiss Medica is a stem cell hospital in Serbia. Our treatment programs are structured to include medical care, diagnostics, and patient support within a single, clearly defined plan, so patients know in advance what is covered. The estimated cost of the stem cell program ranges between €7,000 and €31,000*.
*Prices are indicative and based on 2026 estimates; they may vary depending on condition severity and required cell quantity.
Medical Assessment, Treatment, and Monitoring
The process begins with a remote medical consultation, provided at no cost, to review medical records and determine whether stem cell therapy may be appropriate.
If a patient proceeds, on-site care programs typically include the following medical procedures:
- Review of medical history, current medications, and prior test results
- Diagnostic tests and imaging (performed if the doctor determines they’re necessary for treatment planning).
- Stem cell collection and processing in our own GMP-certified laboratory
- Stem cell injections or infusions according to an agreed protocol
Patients are monitored daily by the medical team, with regular evaluations to track progress and adjust care when needed.
Rehabilitation and Follow-up Care
Rehabilitation therapies are integrated into our program rather than offered as optional add-ons, helping patients regain strength, improve circulation, and support overall health in a safe and supervised way. These may include:
- Physiotherapy and kinesiotherapy to support circulation, mobility, and safe physical activity
- Intracellular Metabolism Recovery (IMR) to support overall metabolic balance and cellular function
- Medical device-based therapies, such as ultrasound, plasmapheresis, and many others, can improve outcomes.
After returning home, patients have access to follow-up communication with the clinic to review progress and share information with their local healthcare providers.
Accommodation and Practical Support
Because many patients travel from abroad, we include practical support to reduce stress and allow patients to focus on their health:
- Accommodation and meals for the patient and one companion
- Airport transfers and local transportation related to care
- Translation support when needed to ensure clear communication
How to Get a Personalized Stem Cell Therapy Cost Estimate
At Swiss Medica, we evaluate each case individually and base our cost estimates on your specific medical details rather than a fixed price list. We begin with a free medical consultation and review of your records, during which our medical team assesses your condition and discusses whether stem cell therapy may be appropriate. Based on this evaluation, we provide a personalized treatment plan with a clear cost outline showing what is included.
Contact us
Book a free consultation to discuss your situation and explore whether stem cell therapy may help. No pressure or commitment.

Medical Advisor, Swiss Medica doctor
Frequently Asked Questions
1. What is the average cost of stem cell therapy?
Most patients pay between $5,000 and $20,000 for a single treatment course.
2. What factors influence stem cell treatment cost the most?
The main drivers are condition complexity, cell type and source, number of injections, level of medical supervision, and clinic location.
3. Does the type of cell used affect the stem cell therapy price?
Yes, autologous cells are frequently expensive due to the obtaining processes, whereas donor cells raise costs due to screening, processing, and storage.
4. How much does a stem cell procedure cost in the US, UK, and Europe?
Typical ranges are $5,000–$50,000 in the US, £4,000–£40,000 in the UK, and €7,000–€50,000+ across Europe, depending on complexity and country.
5. Why do stem cell therapy prices differ between clinics and countries?
Differences stem from labor costs, regulations, taxes, competition, and what services are included in the quoted price.
6. What is included in the stem cell procedure cost?
It varies, but may include evaluation, cell processing, injections or infusions, medical supervision, and short-term follow-up.
7. Are diagnostic tests and follow-up care included in the stem cell therapy price?
Sometimes—they may be bundled or billed separately, so confirmation with the clinic is essential.
8. Are travel and accommodation included in the stem cell treatment price?
Usually no, though some clinics like Swiss Medica include local accommodation and transfers for international patients.
List of References:
Mousaei Ghasroldasht, M., Seok, J., Park, H. S., Liakath Ali, F. B., & Al-Hendy, A. (2022). Stem Cell Therapy: From Idea to Clinical Practice. International journal of molecular sciences, 23(5), 2850. https://doi.org/10.3390/ijms23052850
Wang, Y., Yi, H. & Song, Y. The safety of MSC therapy over the past 15 years: a meta-analysis. Stem Cell Res Ther 12, 545 (2021). https://doi.org/10.1186/s13287-021-02609-x
Zakrzewski, W., Dobrzyński, M., Szymonowicz, M., & Rybak, Z. (2019). Stem cells: past, present, and future. Stem cell research & therapy, 10(1), 68. https://doi.org/10.1186/s13287-019-1165-5



